Fields of Research
Fields of Research
Fields of research of the Industrial Chemistry 2
Our expertise
- Heterogeneous catalyzed gas and liquid phase reactions in flow and gas recycle reactors.
- Heterogeneous catalyzed rearrangement reactions in liquid phase
- Hydroamination of alcohols
- Conversion of methyl butynol for detemination of basic and acidic properties of oxidic catalysts
- Characterization of active centres on heterogeneous catalysts
- Modification of silicious surfaces
- Developement and characterization of mono and bifunctional catalysts on the basis of zeolites, SiO2, Al2O3 and layered silicates.
Characterization of spezialized ion exchanger
- Developement of inorganic-organic composite materials for use in material seperation and catalysis.
-
DRIFT spectroscopy for quantitative analysis of sorption of aromatic amino acids on ion exchangers with styrol divinylbenzene copolymer matrix.
- Investigation of the catalytic activity by methyl butynol testreaction (MBOH).
- Determination of the hydrophilicity of silicalistic materials by hydrophobicity index (HI).
| 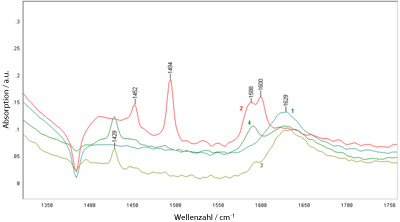 DRIFT measurement |
Developement of new bifunctional catalysts
- With bifunctional reactions hydrogen is activated on the metal and diffuses on the surface to the acidic centre, where the carbon hydrate conversiontakes place (i.e. the hydroisomerization of n-hexane). Due to the mobility of the activated hydrogen it is possible to create the bifunctional catalyst by mixing of the metallic (metal on support) and the acidic component (H-zeolite). The component metal/support can be seperately pre-treated from the zeolite, so that the metal is constrictedly accessible for catalyst poisons, but furthermore active for hydrogen activation.
| 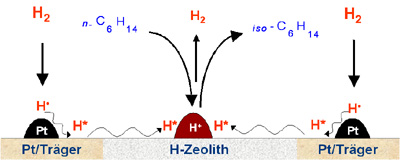 Extended model of bifunctional catalysis |
Synthesis of nanodispersed semiconductors in a zeolite matrix
-
Synthesis of zeolites with CdS and ZnS nanoparticles in the pore structure.
-
DRIFT investigations of the solid state ion exchange on selected examples in different temperature ranges.
-
Temperature-programmed H2/CO evolution for investigation of the chemisorption and the physisorption behaviour, respectively.
| 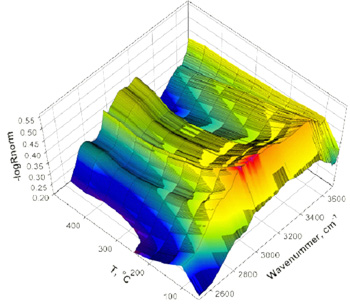 DRIFT measurements of H-beta zeolites subsequent to a H2S treatment |
|
Analytics
-
Surface determination and pore size distribution according to BET.
-
Temperature-programmed reduction (TPR), oxidation (TPO) and hydrogen evolution (TPHE).
-
Temperature-programmed Ammonia desorption (TPAD) for acidic centre characterization.
-
Diffuse reflectance UV/Vis spectroscopy.
-
Differential scanning calorimetry (DSC).
| 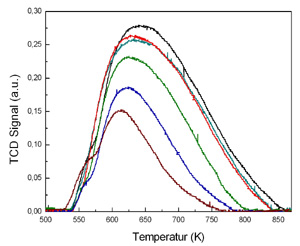 NH3-TPD profile on NaH-beta zeolites |
| |


